- Blog
- Memo template for ms word
- Meal macro planner free
- John dalton-s atomic theory
- Dirty hearts card game online
- Bo1 mod menu ps3 onload usb 2017
- Purble place free game unblocked
- Party planner package
- Cuphead free download for mac
- Desktop clock for windows 10 free download
- Breaking the quiet 3 1080
- Asd in dsm 5
- Invasion de world war ii omaha beach
- Stronghold crusader extreme 1-3-1 e trainer
- Programs better than instant text pro 7
- Download hack pubg for pc
- R studio 8-5 registration key
- Garageband free download for windows 10
- Apk whatsapp download
- Download hindi typing tutor kruti dev
- Amy freeze sexy
- ARC XT PRO vs ARC XT basic
- O negative blood type diet meal plan
- Install windows 11
- Practice math questions for ged
- Snip it tool for pc free download
- Red eyex32 game save bo1 editor download
- Fortnite free code redeem
- Waifu sex simulator futa models
- Time zone map of usa
- Sac state graphic design portfolio
- Arc xt pro
- Rust aimbot esp
- Dj player app for pc free download
- Gamemaker studio 1-4 license key free
- Wooden ring box
- Moca test scoring adhd
- Bible study on elijah
- Kink checklist git
- Spectrum cable tv listings with channel numbers
- Space shuttle endeavour cockpit vr
- Scarlet blade vendetta not opening
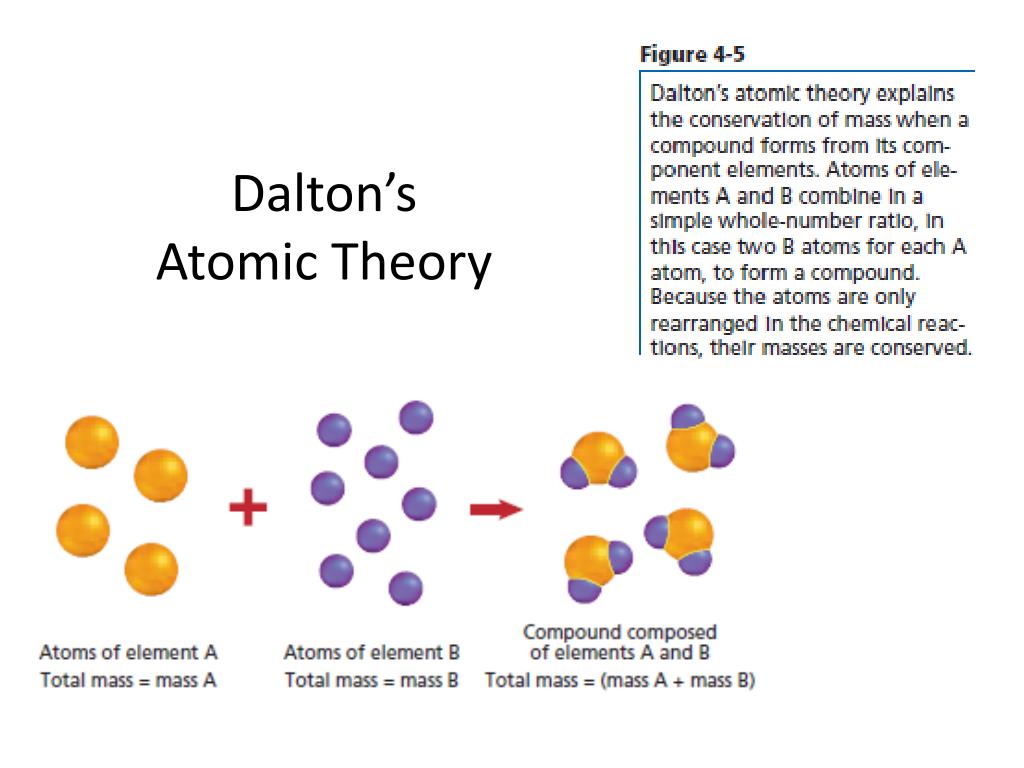
Their masses are different, and their chemical reactions are different. Atoms of different elements have different properties.All atoms of a particular element are identical.The elements are made of atoms, which are tiny particles that are too small to see.He also studied the movement and arrangement of water molecules when they are frozen in ice.Dalton showed what molecules look like when atoms combine to form compounds.From the data he collected, Dalton proposed that matter exists as atoms and that atoms of different elements have different masses.
“If two elements form more than one compound between them, then the ratios of the masses of the second element which combine with a fixed mass of the first element will be ratios of small whole numbers.” In 1808, he published his book, A New System of Chemical Philosophy, in which he made use of countless chemical reactions, stating:.His study of gases made him wonder about the components that make up atoms, which was first proposed by Democritus in ancient Greece.The atomic theory was Dalton’s most significant scientific work in chemistry.“That part of the image which others call red, appears to me little more than a shade, or defect of light after that the orange, yellow and green seem one color, which descends pretty uniformly from an intense to a rare yellow, making what I should call different shades of yellow.”.He proposed that this eye condition is hereditary. This contained the descriptions and observations he discovered in his own brother’s vision. During his stay in Manchester, he was nominated as a member of the Manchester Literary and Philosophical Society, where he initially contributed his first paper on color blindness titled “Extraordinary facts relating to the vision of colors”.John Frederic Daniell acknowledged him as the “Father of Meteorology”.He described partial pressure using a physical law. Dalton measured the air capacity to absorb water vapor and the different partial pressures given the temperature.Being born and raised in the mountainous region of Lake District leveraged Dalton to study and observe meteorological phenomena.It contained essays about meteorology based on his own observations, together with those of John Gough and Peter Crosthwaite. He brought with him the proof sheets of his first book, Meteorological Observations and Essays. In 1793, Dalton left his home and moved to Manchester for a teaching opportunity.In 1787, he started his meteorological diary, which had more than 200,000 observations.Based on records, one of his students enjoyed his way of teaching, recalling: “The boys (were) all glad to be taught by John Dalton, because he had a gentler disposition and besides his mind was so occupied with mathematics, that their faults escaped his notice.”.This lasted until 1793, when he became a math and philosophy tutor at the New College in Manchester.
At the age of 19, Dalton became the school’s principal.These mentors inspired him to teach himself science, mathematics, Latin, Greek, and French.

- Blog
- Memo template for ms word
- Meal macro planner free
- John dalton-s atomic theory
- Dirty hearts card game online
- Bo1 mod menu ps3 onload usb 2017
- Purble place free game unblocked
- Party planner package
- Cuphead free download for mac
- Desktop clock for windows 10 free download
- Breaking the quiet 3 1080
- Asd in dsm 5
- Invasion de world war ii omaha beach
- Stronghold crusader extreme 1-3-1 e trainer
- Programs better than instant text pro 7
- Download hack pubg for pc
- R studio 8-5 registration key
- Garageband free download for windows 10
- Apk whatsapp download
- Download hindi typing tutor kruti dev
- Amy freeze sexy
- ARC XT PRO vs ARC XT basic
- O negative blood type diet meal plan
- Install windows 11
- Practice math questions for ged
- Snip it tool for pc free download
- Red eyex32 game save bo1 editor download
- Fortnite free code redeem
- Waifu sex simulator futa models
- Time zone map of usa
- Sac state graphic design portfolio
- Arc xt pro
- Rust aimbot esp
- Dj player app for pc free download
- Gamemaker studio 1-4 license key free
- Wooden ring box
- Moca test scoring adhd
- Bible study on elijah
- Kink checklist git
- Spectrum cable tv listings with channel numbers
- Space shuttle endeavour cockpit vr
- Scarlet blade vendetta not opening
